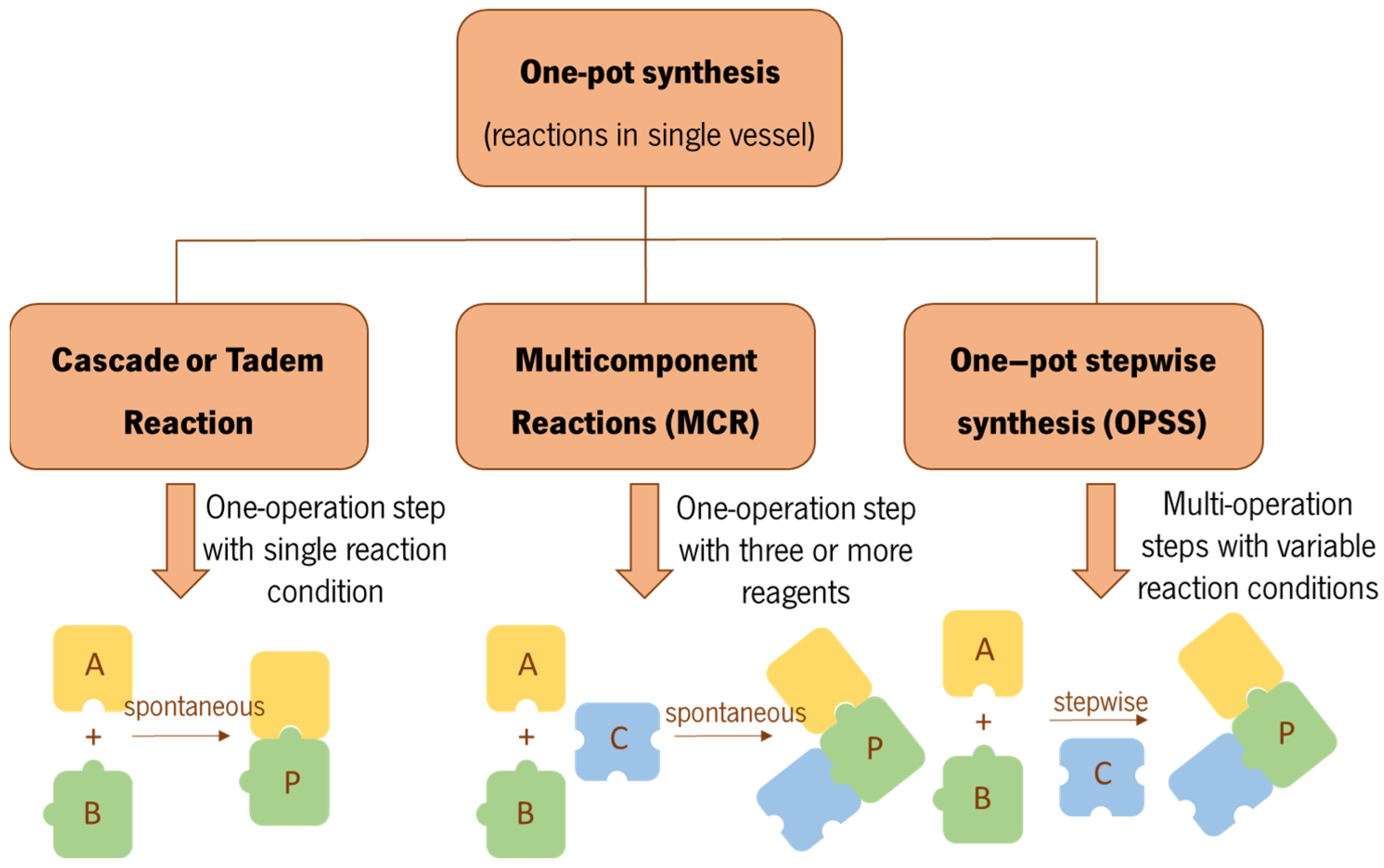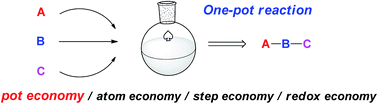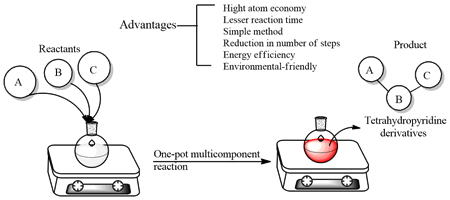
Effect of Methods and Catalysts on the One-pot Synthesis of Tetrahydropyridine Derivatives: A Mini-Review | Orbital: The Electronic Journal of Chemistry
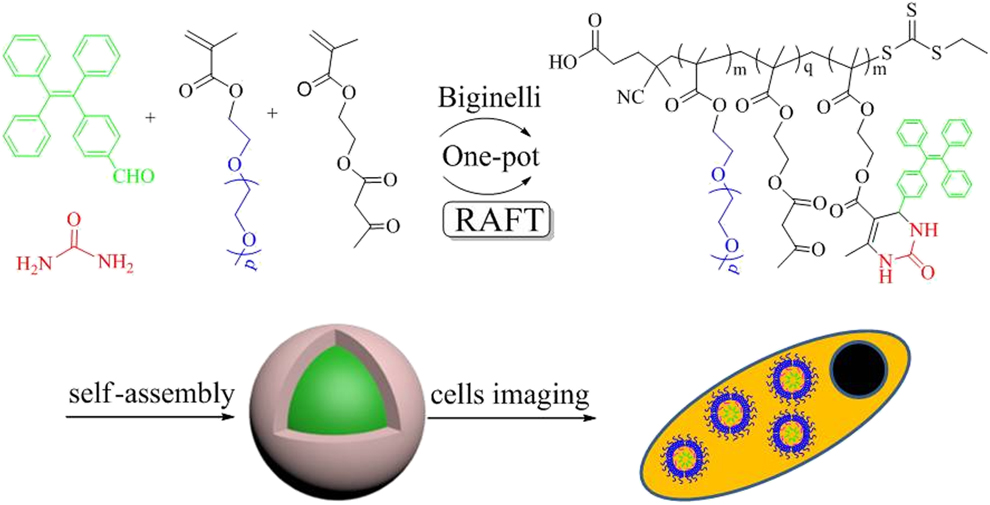
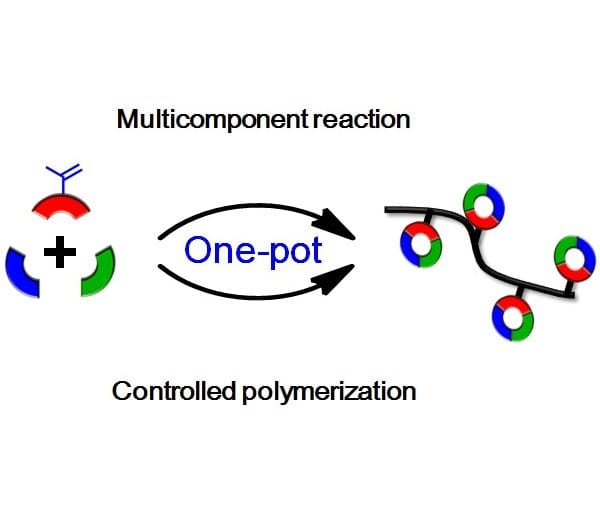
Amphiphilic fluorescent copolymers via one-pot synthesis of RAFT polymerization and multicomponent Biginelli reaction and their cells imaging applications | Journal of Materials Research | Cambridge Core
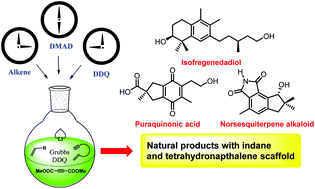
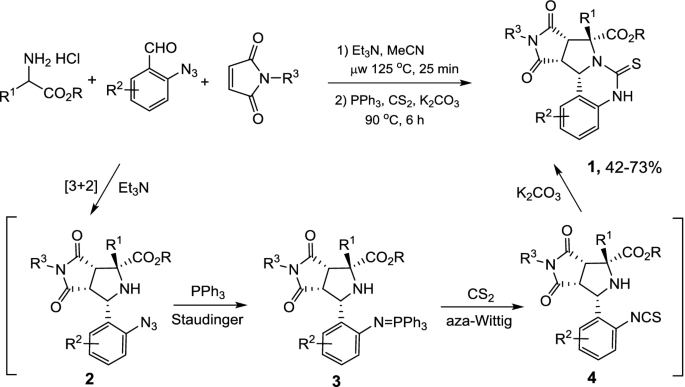
One-pot quadruple/triple reaction sequence: a useful tool for the synthesis of natural products - Organic & Biomolecular Chemistry (RSC Publishing)
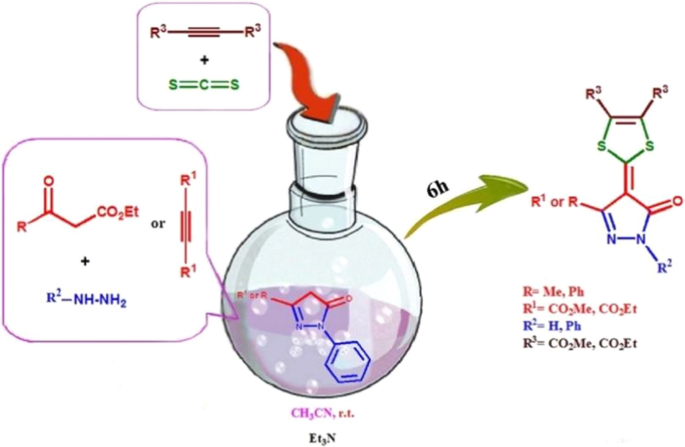
A facile one-pot synthesis of new functionalized pyrazolone-1,4-dithiafulvene hybrids | Molecular Diversity
![One‐Pot Synthesis of Cycloocta[b]indole Through Formal [5+3] Cycloaddition Using Donor–Acceptor Cyclopropanes - Okabe - 2019 - European Journal of Organic Chemistry - Wiley Online Library One‐Pot Synthesis of Cycloocta[b]indole Through Formal [5+3] Cycloaddition Using Donor–Acceptor Cyclopropanes - Okabe - 2019 - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/dadcdf11-c1cf-437b-ba95-879966dd809b/ejoc201900610-toc-0001-m.png)
One‐Pot Synthesis of Cycloocta[b]indole Through Formal [5+3] Cycloaddition Using Donor–Acceptor Cyclopropanes - Okabe - 2019 - European Journal of Organic Chemistry - Wiley Online Library
![One-Pot, Two-Step Synthesis of Imidazo[1,2-a]benzimidazoles via A Multicomponent [4 + 1] Cycloaddition Reaction | ACS Combinatorial Science One-Pot, Two-Step Synthesis of Imidazo[1,2-a]benzimidazoles via A Multicomponent [4 + 1] Cycloaddition Reaction | ACS Combinatorial Science](https://pubs.acs.org/cms/10.1021/co400075z/asset/images/co400075z.social.jpeg_v03)
One-Pot, Two-Step Synthesis of Imidazo[1,2-a]benzimidazoles via A Multicomponent [4 + 1] Cycloaddition Reaction | ACS Combinatorial Science
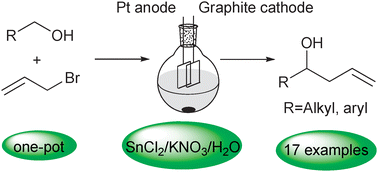
An electrochemical tandem reaction: one-pot synthesis of homoallylic alcohols from alcohols in aqueous media - Chemical Communications (RSC Publishing)

Efficient one-pot synthesis of substituted diphenyl 1, 3-thiazole through multicomponent reaction by using green and efficient Iron-catalyst via Cross-Dehydrogenative Coupling(CDC) | Molecular Diversity

One‐Pot Two‐Step Chemoenzymatic Cascade for the Synthesis of a Bis‐benzofuran Derivative - Mertens - 2019 - European Journal of Organic Chemistry - Wiley Online Library

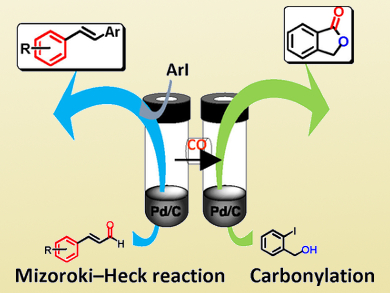
Applied Sciences | Free Full-Text | Sequentially Palladium-Catalyzed Processes in One-Pot Syntheses of Heterocycles
![Development of a Water-Mediated and Catalyst-Free Green Protocol for Easy Access to a Huge Array of Diverse and Densely Functionalized Pyrido[2,3-d:6,5-d′]dipyrimidines via One-Pot Multicomponent Reaction under Ambient Conditions | ACS Sustainable ... Development of a Water-Mediated and Catalyst-Free Green Protocol for Easy Access to a Huge Array of Diverse and Densely Functionalized Pyrido[2,3-d:6,5-d′]dipyrimidines via One-Pot Multicomponent Reaction under Ambient Conditions | ACS Sustainable ...](https://pubs.acs.org/cms/10.1021/acssuschemeng.7b02696/asset/images/medium/sc-2017-02696k_0008.gif)
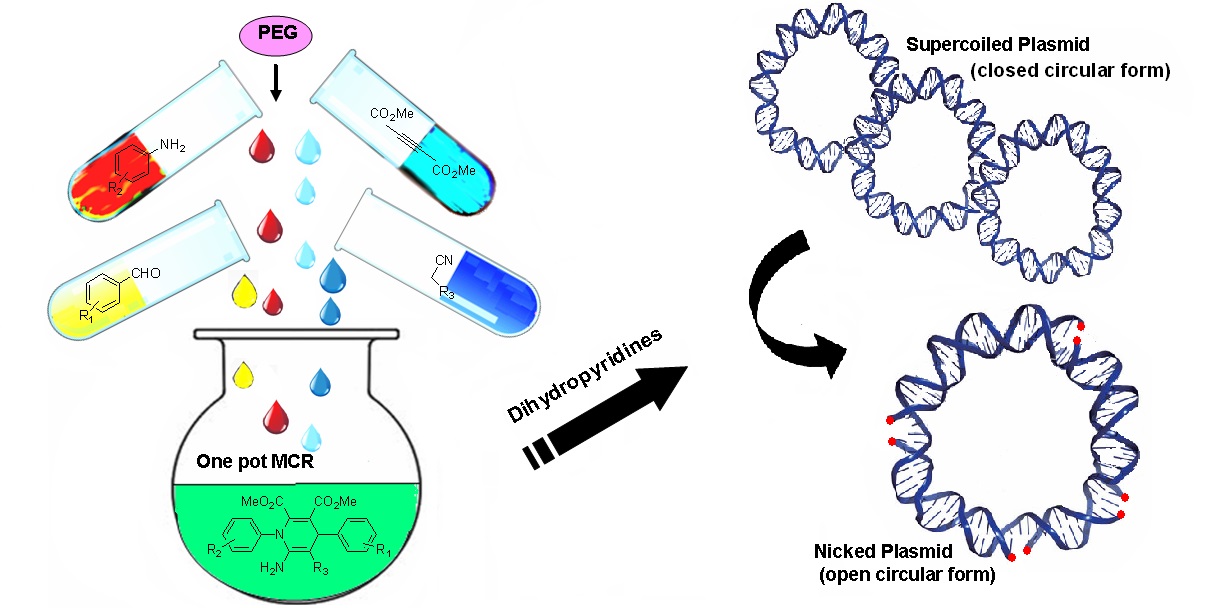
Development of a Water-Mediated and Catalyst-Free Green Protocol for Easy Access to a Huge Array of Diverse and Densely Functionalized Pyrido[2,3-d:6,5-d′]dipyrimidines via One-Pot Multicomponent Reaction under Ambient Conditions | ACS Sustainable ...