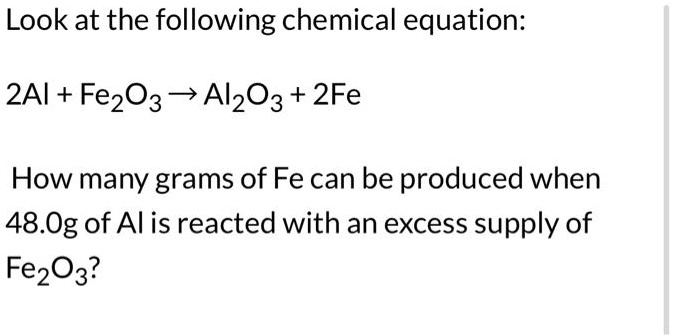SOLVED: Consider the following oxidation-reduction reaction: Fe2O3(l) + 2Al(l) â†' Al2O3(l) + 2Fe(l) Which substance is the oxidizing agent and which one is the reducing agent? Select one: a. Oxidizing agent: Al2O3

4) Name the substance oxidised , substance reduced ,oxidising and reducing agents Also balance the equation wherever necessary a) - Science - Chemical Reactions and Equations - 13670549 | Meritnation.com
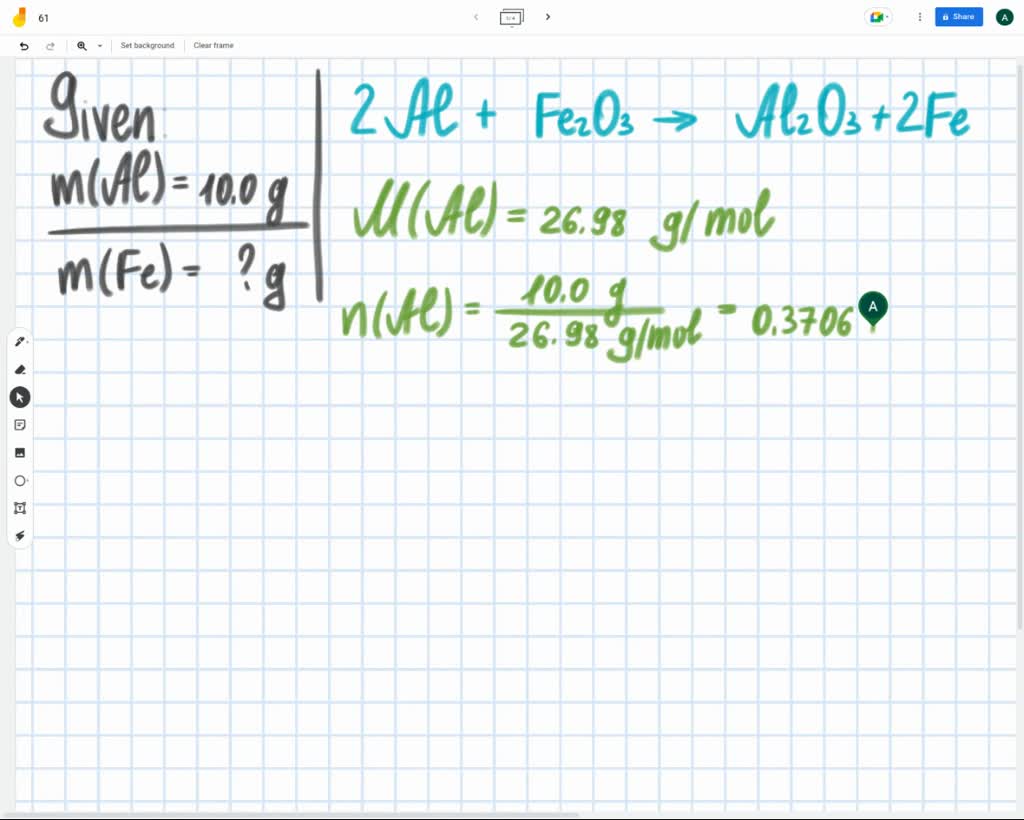
SOLVED: Al + Fe2O3 → Al2O3 + Fe (Need to balance equation) How many grams of Fe can be produced when 10.0g of Al is reacted with an excess (unlimited) supply of Fe2O3?
39. Fe2O3 + 2Al —> Al2O3 + 2Fe Which of the statement about the reaction below are incorrect? (|) Fe is getting reduced. (||)Al2O3 is getting oxidised (

CHAPTER 6 CHEMICAL REACTIONS AND EQUATIONS. Chemical reactions are responsible for just about everything that occurs around us all the time. How do you. - ppt download

SOLVED: Using the balanced equation 2 Al + Fe2O3 = Al2O3 + 2 Fe If you reacted 2.4 moles of aluminum with Fe2O3 and obtained 99.7 g of Al2O3, what would the percent yield of the reaction be?

Al + Fe2O3 → Al2O3 + Fe (Need to balance equation) How many grams of Fe can be produced when 10.0g of Al - brainly.com